Business Sectors
Events
Contents
Why new 0.5% sulphur fuels may produce higher black carbon emissions
Francisco Malta, director/owner VM Industrials - Aderco Australia explains how maritime can learn from the automotive experience of mitigating ‘black carbon’
Recently, I attended the 9th Sulphur Roundtable, organised by the Australian Maritime Safety Authority (AMSA) and the Maritime Industry Australia Limited (MIAL).
The purpose of these roundtables is for those in shipping who are impacted by the IMO 2020 sulphur limit changes to understand the new rules and regulations and to have a voice in IMO discussions.
As a whole, Australia and shipping have managed the sulphur cap transition relatively well, with only minor glitches and without too much disruption.
One topic raised at the above-mentioned roundtable involved a concern in the possibility of an increase in black carbon (BC) emissions, a topic that has come to the fore since the introduction of very-low sulphur fuel oil (VLSFO - 0.5% sulphur). It is particularly noticeable in the Arctic, with its snow-white landscape.
The Clean Arctic Alliance (CAA) has called for immediate action to stop soot landing on ice, as the soot retains heat from the sun and speeds up the melting of the Arctic.
IMO (SUB-COMMITTEE ON POLLUTION PREVENTION AND RESPONSE 7th session Agenda item 8) made a claim that the new VLSFOs are blends with a high content of aromatics hydrocarbons and that this was the reason behind the BC emissions
However, we have found VLSFOs are actually comprised of more paraffinic fuels. The International Bunker Industry Association (IBIA) has also debunked IMO’s aromatic theory and confirmed the majority of new O.5% fuels are mostly paraffinic and should emit less BC emissions.
The realisation of this phenomenon dates back to the automotive diesel-gate fiasco of 2011.
As countries in the western world began gradually reducing sulphur limits for fuels used in vehicles, a change came about in the method to ’economically’ produce ultra-low sulphur fuels (ULSFs), with as little as 10 and 15ppm (0.01%-0.015%) in sulphur content.
These fuels were no longer refined in one location and delivered to the fuel supplier or fuel user. That was too costly. A study prepared for The International Council On Clean Transportation (ICCT) in 2012 concluded the cost to de-sulphurise fuel at a single refinery was anywhere between US$0.8 cents and US$3.2 cents per litre.
The 2012 ICCT study stated: “The estimated national average costs of producing 10 ppm ULSD (in order of increasing costs) are in the ranges of 0.8¢ to 1.1¢/liter (India), 1.7¢ to 2.2¢/liter (China), 2.0¢ to 2.7¢/liter (Brazil) and 2.5¢ to 3.2¢/liter (Mexico).”
Where did things go wrong?
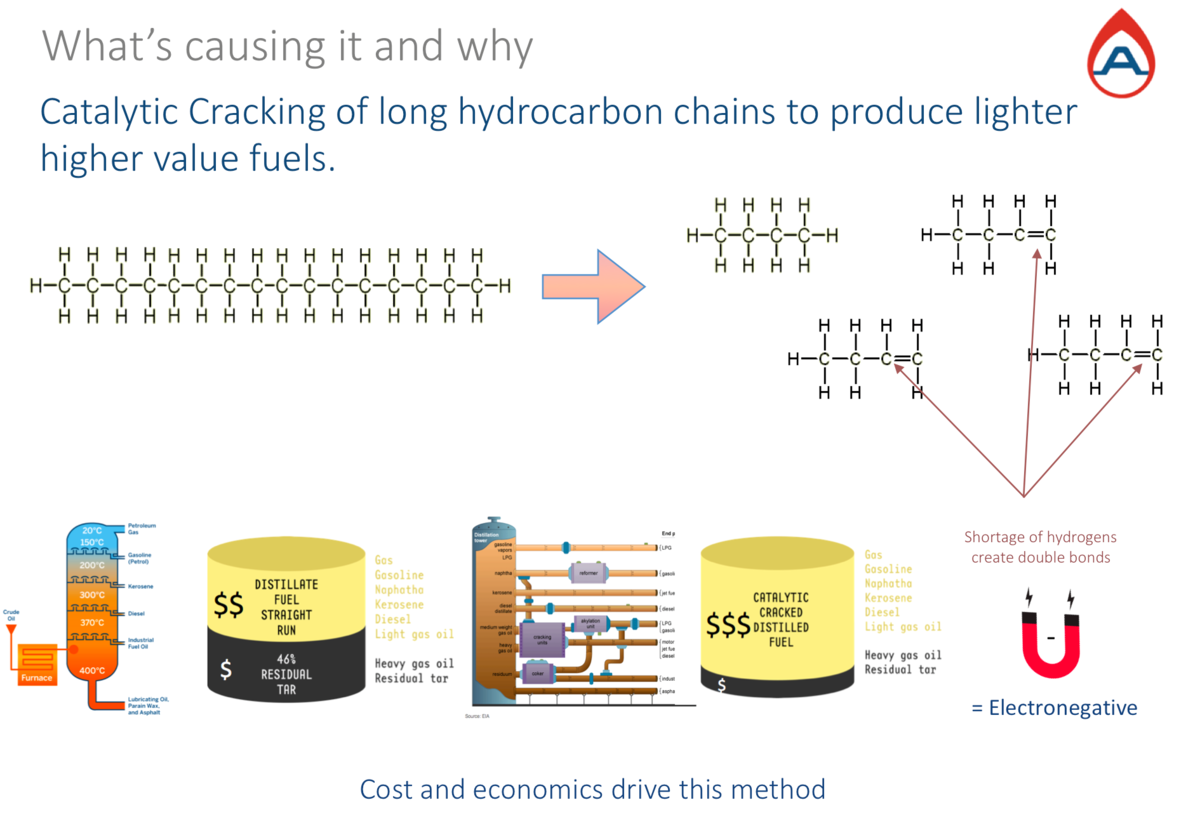
The image above shows a hydrocarbon chain fractioned into many smaller chains to produce lighter fuels with a higher value from a barrel of crude oil. These processes are not new in refining; they have been tried and improved over many years, dating back to the beginning of refining fossil fuels; however, they play an important role in the issue today.
Notice on the left of the image, the hydrocarbon chain is completely circled by hydrogens and as the chain is fractioned (as seen on the right) there are not enough hydrogens to completely encircle the smaller chains.
In a controlled environment, the fractioned chains are hydrogenated; this means adding the missing hydrogen molecules to avoid the double-bond effect seen on the right with the pointing red arrows. When hydrogenated, the fractioned chains are re-stabilised.
This would be the normal process in a refinery without blending.
However, if hydrogenation does not take place the fractioned chains remain double-bonded, incomplete, and unstable. Their electronegativity increases and the chains become magnetic; they attract other hydrogens from other chains and hydrogens from water (H2O) droplets, present in fuels from condensation.
Unfortunately, when blending batches from many different refineries and sources this process is no longer taking place on every batch. As this is at a molecular level and does not change the fuel specification, the batches with a shortage in hydrogens are not identifiable.
The other negative factor with double bonds is their bonds have twice the strength and therefore require twice the energy to combust.
So, in simple terms, the fuels are now magnetic and holding on to other hydrocarbons and water droplets - remembering that water is made of two hydrogen atoms and one oxygen atom.
The result is a far heavier agglomeration of multiple paraffinic chains with water, requiring far more energy to combust. Therefore, the combustion results in unburnt hydrocarbons that exit as black smoke emissions.
In modern cars, BC emissions are not visible because most cars have diesel particulate filters (DPFs) to capture these un-burnt particles; but these have been problematic, with car DPFs filling up more rapidly than they could regenerate.
As the unstable hydrocarbons continue to agglomerate, the fuels become heavier than they were intended to be; they continue to be ‘on specification’ but complete combustion is now far more difficult for an engine to achieve.
Back to shipping
The irony is that HFO 3.5% S containing asphaltenes often have a number with highly electronegative hydrocarbons that attract and agglomerate other asphaltene molecules. The difference is that when these agglomerate they become so heavy they drop from suspension and end up as sludge in tanks; they rarely make their way to combustion unless singular.
In this new VLSFO scenario, the agglomerations of paraffinic molecules remain light enough and suspended and do make their way to the combustion chamber.
Some car manufacturers resigned to fitting devices that switched off the exhaust gas recirculation (EGR) allowing an increase in burn temperatures (therefore emitting higher nitric oxides) to achieve the temperature required to fully combust.
However, as these practices became exposed, many had no option but to give up on diesel and start to plan for an electric future.
The impact ULSF blending would have on the automotive industry was not understood at the time; it is now fairly clear and confirmed, as every major fuel supplier offers special premium fuel as a solution.
These premium-fuel solutions come in different forms of additives, from solvent-based combustion improvers that alter fuel specifications to reach complete combustion, to non-solvent green technologies that physically neutralise the electronegative effect without altering the fuel specifications.
In all cases, the desired outcome is complete combustion, without leaving the excess in partially burnt fuel resulting in BC emissions.
BC emissions are most prevalent at low engine speeds when combustion is most difficult. This may explain why so much is present in the Arctic; not only is the black on the white landscape more visibly obvious, it is likely ships are operating at low loads with caution to navigate icebergs. This would not be exclusive to the Arctic; given the same fuel, engine and operating conditions, BC could happen in any location.
This mirrors the automotive world (albeit without the icebergs!) and explains why cars in cities were most impacted; they were operating at low engine loads and speeds.
Conclusion
Blending is here to stay, there is no turning back. Blending is the only economical method to produce low-sulphur fuels and just like the automotive industry, the shipping industry may not be able to operate without excessive BC emissions if fuel stability is not addressed.
But thanks to the automotive industry, shipping now has the advantage of understanding the problem and having a better idea of how to fix it.
References
IMO - imoarcticsummit.org/wp-content/uploads/2020/02/PPR-7-8-Initial-results-of-a-Black-Carbon-measurement-campaign-with-emphasis-on-the-impact-of-the...-Finland-and-Germany-1.pdf
IBIA - ibia.net/black-carbon-misunderstandings-thoroughly-discussed-and-addressed/
ICCT - theicct.org/publications/technical-and-economic-analysis-transition-ultra-low-sulfur-fuels-brazil-china-india
CAA - www.hfofreearctic.org/en/2018/01/24/infographic-can-reduce-black-carbon-emissions-international-shipping/
Related to this Story
Events
Maritime Cyber Security Webinar Week
International Chemical & Product Tanker Conference 2024
Marine Propulsion: Fuels Webinar Week
How enhanced connectivity is propelling maritime into the AI era
© 2023 Riviera Maritime Media Ltd.